Kenyan Bishops not Satisfied with Government Response to Vaccine Controversy
The concern that the neonatal tetanus vaccines in Kenya sponsored by the World Health Organization are laced with sterilizing agents first arose months ago, and the uncertainty continues as the newest round of testing produced another inconclusive standoff between the Catholic Health Commission of Kenya and the Ministry of Health.
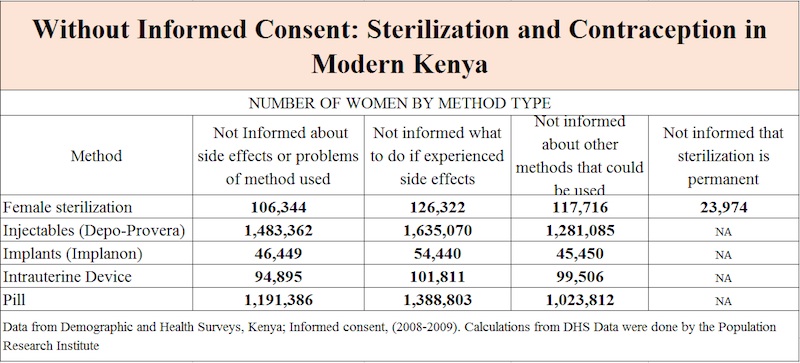
The Kenyan Bishops initially expressed concern in March and again in October of 2014. The neonatal tetanus vaccine, which is normally given to women of reproductive age, was being given to women in five doses, rather than the usual two or three doses. Given past abuse regarding informed consent and sterilization in Kenya (see table), the extraneous vaccinations provoked concern. The Catholic Church requested the Kenyan Ministry of Health to test the vaccines for the presence of sterilizing agent, beta HCG. After the Health Ministry declined to test the vaccines, the Catholic Church in Kenya acquired nine vials of the vaccines and tested six vials in five different Kenyan laboratories.
Three of the these vaccines returned positive for Beta HCG.[1]
The Ministry of Health stated that they had separately tested the tetanus vaccine but found the vaccine to be safe.
After ensuing public outcry and confusion, Kenya's Ministry of Health and the Catholic Church formed a joint committee in of experts in November to jointly test the vaccines. The Catholic Church resubmitted their nine vials.
The Ministry of Health did not have any samples from their earlier testing, so they acquired new samples from the field and central store with the same batch number of the other samples. The Ministry of Health submitted 50 such vials.
A private Kenyan laboratory, AgriQ Kenya, tested the 59 samples (50 from the Ministry of Health, and 9 from the Catholic Church) with a method called High Performance Liquid Chromatography. High Performance Liquid Chromatography separates components of a given mixture using a pressurized liquid solvent and is commonly used to test hormones, poisons, and pesticides. Of the 59 samples tested, 56 returned clean; the same 3 vials which had tested positive for Beta HCG previously, returned positive again.
Although the presence of Beta HCG in the three vials is now undisputed, officials on both sides are concerned about how the Beta HCG made its way into neonatal tetanus vaccines.
After the testing at the Kenyan laboratory, the director of Medical Services, Nicholas Muragrui endorsed the tetanus program , saying, "the immunisation campaign in Kenya is safe and the tetanus campaign should continue."
The Chairman of the Kenya Conference of Catholic Bishops, John Cardinal Njue, issued a statement saying, "the vaccines used for the mass tetanus vaccination campaigns in March and October 2014 contained some vials laced with Beta HCG. It is worth noting that only the Catholic Church had collected samples during the actual March and October 2014 tetanus vaccination campaigns."
The statement of the Kenya conference of catholic bishops concluded by saying, "We insist that no further mass tetanus vaccination campaigns should be undertaken in Kenya before the vaccines have been appropriately tested and proven to be safe."
10 million of Kenya's 45 million residents are Catholic, and the health sector and the Kenyan church are well integrated.
The Chairman of the Catholic Health Commission of Kenya affirmed their participation in health care saying, "Health service delivery forms an integral part of evangelization of the Catholic Church.
While the Kenyan church views health ministry as an "integral part" of their apostolic mission, the Church's health ministry is also integral to Kenya's overall health infrastructure. According to recent estimates , the Catholic Church in Kenya operates one tenth of the nation's hospitals and health centers, and an eighth of all dispensaries.
Considering that three of the vials were confirmed by two labs to contain a sterilizing agent in the supposedly simple neonatal tetanus shot, Kenya should use a different provider for vaccines until the investigation is resolved. Even one woman sterilized without consent is one too many.
.
Endnote:
[1] Three of the six vaccine samples tested contain significant levels of human chorionic gonadotropin (HCG):
- Sample KA retention time (RT) 36.283 and peak area of 37.539%
- Sample KB retention time (RT) 35.825 and peak area of 26.512%
- Sample KC retention time (RT) 38.042 and peak area of 23.939%
Article copyrights are held solely by author.
[ Japan-Lifeissues.net ] [ OMI Japan/Korea ]