[Book] Breast Cancer: Its Link to Abortion and the Birth Control Pill
Essential Background
Over the past few decades, researchers have noted that the normal development of a woman's breast depends on a number of factors. Central factors in this process include two classes of female sex hormones called estrogens (estrogens are spelled oestrogens by the British) and progestins and a pheromone (ie, similar to a hormone, but is secreted by one member of a species and affects another member) called hCG (human Chorionic Gonadotropin). In the human body, estradiol (which is a type of estrogen) and progesterone (which is a specific type of progestin) are referred to as the dominant sexual hormones. The breast requires a proper level and balance of both of these hormones if it is to develop normally. However, when the breast is exposed to synthetic estrogens and⁄or progestins, or to a rapid fall in hormone levels, it may be at risk of becoming cancerous as noted in animal studies [1, 2, 3, 4, 5].
Oral contraceptives are composed of a synthetic estrogen⁄progestin combination and put the breast cells of a young woman at risk, because these hormones increase the rate of cell division, a process called mitosis [6, p.375]. In general the higher the mitotic rate of a cell, the higher the risk that it will turn into a cancer cell.
In regard to abortion, when a woman becomes pregnant she experiences a dramatic increase in her hormone levels of estradiol, progesterone, and hCG. The initial increase in hormone levels induces breast cells to divide and undergo a maturing process called differentiation, which lasts throughout a woman's pregnancy and is completed only after her first term baby is delivered. Hormone levels fall rapidly if she has an induced abortion, leaving her breast cells in a transitional state where they have not completely matured (differentiated) and are vulnerable to carcinogens (ie, factors that cause cancer).
On this note, a number of researchers have studied the effects of having an abortion at a young age and⁄or taking oral contraceptive pills (OCPs), in relation to an increased risk of developing breast cancer. Dr. Emily White, a researcher from Washington State, observed in 1987: “Recently, two other factors have emerged as possible risk factors for breast cancer: oral contraceptive use before first pregnancy and abortion before first term pregnancy.” She adds, “Exposure to induced abortion and oral contraceptives has increased dramatically” [7, p.242]. Let us now explore in more detail the background of these risk factors and the settings in which they influence a woman's risk for developing breast cancer.
Q–1A: What happens to the breast during pregnancy?
The anatomy of the normal breast is depicted in Figure 1A. The mature breast consists of about 15 to 25 alveolar (ie, grape–like) glands, called mammary lobules, which drain into the lactiferous ducts. These in turn empty the mother's milk through small pores in the areola — the pigmented area of skin which surrounds the nipple.
During a woman's pregnancy, her breasts are influenced by many hormones including prolactin, cortisol, insulin, human Chorionic Gonadotropin (hCG) and others, but the main three hormones are estradiol, progesterone, and hCG. These particular hormones reach levels that are far higher than those experienced by a woman at any other time of her life and cause a particularly rapid growth and maturation of the breast cells, especially during the first trimester of pregnancy. This is why women note breast tenderness and swelling during pregnancy. The combination of the high levels of estradiol, progesterone, and hCG during pregnancy serve to help complete the maturation process in the latter half of a woman's pregnancy.
Figure 1A: Normal Breast Anatomy
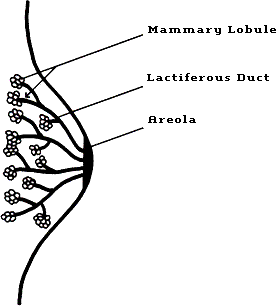
Figure 1B compares two stages of a woman's breast. The first, is the virginal stage, which is found in women who are nulliparous (ie, they have not borne children). Here one notes that most of the ends of the ducts, called terminal ducts, are still immature and have not sprouted into fully formed lobules as noted in Figure 1B–2. These terminal ducts contain many more immature or undifferentiated cells than are found in the mature lobule.
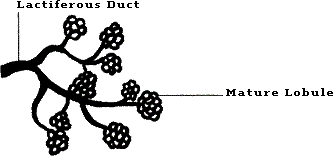
After a woman delivers her first child, we can see in Figure 1B–2, that the breast has formed many mature lobules. These mature cells are more protected from carcinogenic (ie, cancer producing) influence because many of them are fully differentiated (matured). Most cancers begin in immature terminal ducts in cells that are not fully differentiated.
Figure 1B1: Breast Development (Early)
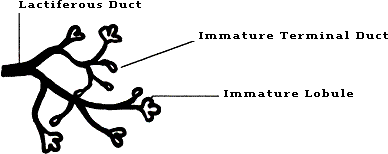
Figure 1B2: Breast Development (Late)
Q–IB: What is breast cancer?
What is it about the cells of a woman with breast cancer that make them different from normal breast cells? The main difference is that breast cancer cells tend to grow rapidly and uncontrollably. Normal breast cells will stop growing when the area in which they are growing gets too crowded. Breast cancer cells continue to grow in an uninhibited fashion even when they have little room to continue to grow. Breast cancer then is composed of millions of breast cancer cells that literally do not know when to quit growing.
Q–IC: Where did the breast cancer cells come from?
Breast cancer cells actually came from normal breast cells through a process called carcinogenesis. Normally, cells divide and grow by a process called mitosis. During this process the original cell divides, resulting in two cells with the same nuclear material as the original cell. The nucleus contains the chromosomes that in turn contain the genes. The chromosomes are composed of strands of genetic material called DNA (deoxyribonucleic acid). During the mitotic process a “mistake” may occur in which one of the cells gets damaged DNA, often because of the influence of a carcinogen, such as radiation or certain chemicals called “super–oxides.” These injuries may result in an abnormal cell — that is, one that tends to grow and divide much more rapidly than normal breast cells.
The initial factor that starts the process of changing a normal breast cell into a cancerous one is called an initiator. The factors which continue the process of converting the now changed cell into a full blown cancer are called promoters. Unfortunately, the original cancer cell divides rapidly, forming a mass or cluster of abnormal cells. After enough time elapses, these cells will form into a mass that is large enough to be felt or detected by mammography (clinically, cancer usually feels like a rock in the breast). The time between the formation of the original abnormal cancer cell and the point at which it has divided and grown large enough to be felt as a lump is usually several years.
The cells of a woman's breast depend on a number of hormones to complete their maturation (differentiation) process. The principal ones are estradiol, progesterone. and especially hCG (human Chorionic Gonadotropin). The breast cells undergo the greatest amount of maturation when a woman's first full–term pregnancy (FFTP) is completed, as was noted in Figure 1B2. Two prominent cancer researchers, Jose and Irma Russo, have demonstrated this phenomenon in their classic work, both in the rat model as well as in humans [1, 2, 8]. In their research, the Russos meticulously documented the microscopic changes that occur in both the rat and human breast during the various stages of life, specifically, before, during and after pregnancy and lactation. They state: “The human breast undergoes a complete series of changes from birth to senility but the differentiation process only takes place through pregnancy and lactation.” [1, p.56].
Q–ID: What factors are known to cause or influence the risk of breast cancer?
There are several factors that cause breast cancer (either by initiation or promotion) or increase a woman's tendency toward it (eg, having a strong family history of breast cancer). The following list includes most of the known risk factors and/or causes of breast cancer:
Note: Often people get confused with the concept of “increased risk.” If a particular risk factor increases the risk of getting breast cancer for a group of women by 50%, it does not mean that 50% of that group of women will get breast cancer. It means that the group of women in question will have a 50% higher incidence of getting breast cancer than the general population. If the general population of women in the U.S. has a lifetime breast cancer risk of 12.5%, a 50% increased risk would mean that the affected group would have an 18.75% overall lifetime risk. A 100% increased risk would mean a 25% lifetime risk.
Age:
In the U.S., and in most parts of the world, the incidence of breast cancer increases dramatically as a woman ages. The table below based on data from the National Cancer Institute (1997), shows the percentage of U.S. women who get breast cancer in the various age groups [9, p.9].
| Age | 1-39 | 1-59 | 1-79 | Lifetime Risk |
|---|---|---|---|---|
| Percent of women who get breast cancer | 0.47% (1 out of 213) | 4.38% (1 out of 23) | 11.31% (1 out of 9) | 12.64% (1 out of 8) |
|
Source: Cancer Statistics, 1996. American Cancer Society. |
||||
Family history:
In general, women who have a first degree relative (eg, a mother or a sister) who has or had breast cancer, have a 2– to 2.5–fold greater risk of developing breast cancer [10, p.2127]. This risk increases to a 4–fold risk if the particular relative developed the cancer before menopause or in both breasts (ie, bilateral breast cancer) [11].
Country of birth:
The rates of breast cancer vary tremendously from country to country. For example, the U.S. has at least a 5–fold higher rate compared to countries such as Iceland or Japan. Dr. Kelsey wrote: “Low rates are found in most Asian and African countries, intermediate rates in southern European and South American countries, and high rates in North American and northern European countries.” [11, p.75] It is interesting to note that when a group of people migrate from a country with a low rate of breast cancer to a country with a high rate such as the U.S., the original group will begin to approach a similar incidence of breast cancer as is in the country to which they migrated after living there for two or three generations. For example: “Native Japanese women who migrate to the U.S. as young adults have a small increase in their breast cancer rates while living in the U.S., whereas Japanese women born in the U.S. have rates approaching those of their white counterparts” [12, p.S11–12]. This indicates that there are certainly factors in addition to genetics that influence a woman's risk of developing breast cancer.
Estrogens in general:
For a long period of time it was thought that estrogens were the only female hormone that increased a woman's risk of breast cancer. As noted below (under progestins), this is no longer the case. Nevertheless, estrogens are still considered to play a major role. It became obvious over the years that women who experienced less “total estrogen exposure,” either because of an early hysterectomy with removal of the ovaries, or because of late onset of menarche (ie, the start of menses) or early onset of menopause, all had a decreased risk of getting breast cancer. In general, women whose bodies experience more exposure to estrogens, whether natural or synthetic, have a higher risk of breast cancer than women who experience less exposure.
Progestins in general:
Twenty years ago, most researchers paid little attention to progestins as a major factor in the development of breast cancer [11], yet today, few would disclaim their role as a significant factor. Progestins seem to increase the risk of breast cancer at least as much and possibly more than estrogens. Animal studies have shown that progestins increased the risk of breast cancer in rodents and dogs [13, 14]. Experiments by the World Health Organization (WHO) on women around the world showed that women who had been injected with long–lasting progestin contraceptives such as Depo–Provera (depot–medroxyprogesterone acetate) for at least 2 years before the age of 25 had a 190% or more increased risk of developing breast cancer [15]. Furthermore, Anderson et al have shown that the rate of cell division in the breasts of nulliparous women (ie, women who have not borne children) is highest in the phase of the menstrual cycle in which progesterone levels are highest [1, p.1140].
Induced abortion (in young women):
There have been several prominent studies which show that women who had an induced abortion at a young age have an increased risk of developing breast cancer. A team of researchers (Russo and Russo) have also shown that rats who had a surgical abortion before giving birth or those who were never pregnant (ie, virgin rats), were both at far higher risk of getting breast cancer than rats who gave birth, when all groups were exposed to the same carcinogen (ie, cancer producing agent) [2]. The most prominent and meticulous meta–analysis* in the medical literature on breast cancer risk due to induced abortion was performed by Brind, Chinchilli, Severs and Summy–Long in 1996. These investigators reported a conservative increased risk of 50% to women who had had an induced abortion prior to their first full–term pregnancy (FFTP) and a 30% increased risk for women who had an induced abortion after their FFTP [16].
*A meta–analysis is a particular type of study which pools together the results of most of the major studies in a field of medicine and then gives an overall risk for the risk factor in question based on the collective data and/or results of those studies.
Oral contraceptive use at a young age:
“Several studies have suggested that the use of oral contraceptives in the early teen years may increase the risk of subsequent breast cancer.” [Haskell 17, p.327]. Obviously the focus of this book will be to explain why, and how much, early use of oral contraceptives contributes to the development of breast cancer. There have been a number of meta–analyses [18, 19, 20, etc.] that have tried to measure the risk of using oral contraceptive pills (OCPs) on breast cancer, but unfortunately they do not always test for the risk in women who used them prior to their FFTP.
For example, the largest meta–analysis done to date, a study that was published both in The Lancet and in Contraception, had no data on the risk of contraceptive use in women who used them prior to their FFTP. (For a detailed analysis of this study see Appendix 4 [of book].) The last metaanalysis which specifically examined the risk of OCPs to women who used them for 4 or more years prior to their FFTP was done by Dr. Isabelle Romieu in 1990 [18]. She noted that women who had taken OCPs for 4 or more years prior to their FFTP had a 72% increased risk of developing breast cancer [RR (relative risk)=1.72 (1.36-2.19)]. (See Chapter 4, question 4D, for a definition of the term “relative risk.” -- Book: Breast Cancer, It's Link to Abortion and the Birth Control Pill by Chris Kahlenborn, MD.)
Miscarriage:
Several studies have noted an increased trend or risk of developing breast cancer when a woman has a miscarriage before her FFTP as noted in Table 1B. Further research or a meta–analysis is urgently needed to verify if this is indeed a true risk factor.
| Author | Year of Publication | Percent Change | Confidence |
|---|---|---|---|
| Pike et al [21] | 1981 | 151% increase(A) | unknown |
| Brinton [22] | 1983 | 9% increase* | 0.8–1.5 |
| Hadjimichael [23] | 1986 | 250% increase* | 1.7–7.4 |
| Rosenberg [24] | 1988 | 10% (decrease)* | 0.7–1.4** |
| Edwertz⁄Duffy [25] | 1988 | 163% increase* | 0.83–8.32*** |
| Adami [26] | 1990 | 20% increase* | 0.7–2.0 |
| Daling [27] | 1994 | 10% (decrease)* | 0.6–1.3 |
| Rookus [28] | 1996 | 40% increase* | 1.0–1.9 |
|
|
|||
It should be noted that women who have miscarriage after their first full–term pregnancy do not seem to have significantly increased risk of developing breast cancer.
Menopause:
"It has been estimated that women whose natural menopause occurs before age 45 have only one-half the breast cancer risk of those whose menopause occurs after age 55." [12, p.S13]
Menarche (ie, the start of a woman's menstrual cycles in life):
A number of studies have noted that having a later onset of menses confers a small decrease in breast cancer risk — usually about 20 to 25%, although many of these results are not statistically significant [27, 29, 30, 31, 33, 34]. Of note, young women who are obese and⁄or sedentary tend to have an earlier age of menarche, whereas women who exercise consistently tend to delay the age of menarche.
Parity (refers to how many children a woman has):
Several studies have noted that the risk of breast cancer decreases as a woman has more children. In general, women who have more children have a lower risk of breast and ovarian cancer. Women who have five or more children have about a 50% decreased risk of breast cancer [34, 35, 36, 37, 38, 39, 40]. In contrast women who are nulliparous have about double the risk of attaining breast cancer compared to the general population [11].
Age at first birth:
Researchers have noted that the risk in mothers who give birth at a young age (ie, under the age of 20) is about one–half to one–third of that of women who deliver their firstborn child after the age of 35 [34, 36, 37, 39, 40, 41]. The main reason for this is that when a younger woman has a child the cells in the small ducts of her breast mature (differentiate) and are less susceptible to cancer producing agents (carcinogens) [see Figure 1B].
Certain types of fibrocystic disease:
Many women have been told that they have “fibrocystic breasts” or “fibrocvstic breast disease.” There are four types: 1) fibrosis; 2) cyst formation; 3) sclerosing adenosis; and 4) duct epithelial hyperplasia. A woman is said to have duct epithelial hyperplasia if a biopsy of one of her breast nodules reveals “hyperplastic cells” — that is, cells which proliferate more abundantly than normal breast cells. Sometimes these hyperplastic cells display “atypical features” when viewed under a microscope, that is atypical hyperplasia. Dr. Colditz, from Harvard, noted that atypical hyperplasia is associated with a 3.7 fold risk of developing breast cancer [42]. Henderson [10] noted that women with atypical hyperplasia had a 4.4–fold risk, which increased to almost a 9–fold risk if a woman also had a family history of breast cancer.
Previous personal history of breast cancer:
The cumulative lifetime incidence rate of a second breast cancer occurring in women who have already had one breast cancer has been reported to be about 25% [10].
Laterality:
Most investigators have noted that breast cancer is found about 10% more often in the left breast than in the right [11]. This may be due to a higher tendency to breastfeed with the right breast.
Long–term breastfeeding:
In a large study, Newcomb et al [43] noted that premenopausal women (ie, women who have not experienced menopause yet) who breast fed for more than 4 months in their lifetime, had at least a 22% decreased risk of breast cancer, whereas postmenopausal women showed no obvious benefit from a history of breastfeeding [43]. In a study of 329 women, McTiernan et al [44] noted that premenopausal women who breast fed for more than 13 months had a 55% decreased risk, whereas postmenopausal women who did so [in their reproductive years] had a 62% decreased risk. Byers et al [45] also noted a decreased risk of breast cancer in premenopausal women who breastfed, but failed to find it in postmenopausal women. Wang also noted that women who breastfed more than 6 years had a 60% decreased risk. Mayberry et al [46] noted that parous black women (ie, black women who have had children) who breastfed for more than 16 months had half the risk of breast cancer compared to black women who breastfed less than 4 months. In general, breastfeeding (lactation) appears to reduce the risk of breast cancer in premenopausal women but further studies are needed to assess how much benefit it has in postmenopausal women.
[In addition, for researchers: It should be pointed out that prolonged lactation (ie, greater than 25 weeks) results in a 40% reduction [RR=0.6 (0.3–1.0)] of the risk of p53+ tumors [47] (a decrease in the p53 [over expression] level means an increase in the amount of tumor suppressor gene, which is beneficial)].
Postmenopausal hormone use (ie, hormone replacement therapy [HRT]):
Most major studies show that women who take artificial estrogen and⁄or progestin for a number of years after menopause have a significantly increased risk of breast cancer. Yang, Daling and colleagues [48] noted that women who had taken estrogen for more than 10 years had a 60% increased risk 1.6 [1.1–2.5] of breast cancer. Colditz et al in a study of 1,935 women who developed breast cancer, noted that women who were currently taking hormonal therapy had a 41% increased risk in getting breast cancer [RR=1.41 (1.15–1.74)] and that women who were taking progestins alone had a 124% increased risk [RR=2.24 (1.26–3.98)] [49, p.1590]. Finally, in an extremely large meta–analysis Beral noted that women who had used estrogen replacement within the last 4 years had a 35% increased risk of developing breast cancer if they had taken the estrogen for 5 years or more [50].
Religion:
Jewish women seem to have a 2.8–fold increased risk of getting breast cancer, perhaps because of their higher rate of the defective BRCA1 gene and⁄or environmental factors [36].
Alcohol:
Longnecker et al [51] in his meta–analysis in 1994 noted that women who drank about two drinks a day had a 25% increased risk of breast cancer (“one drink” would be the equivalent of one beer or one mixed drink or one 4 oz. glass of wine). His large study in 1995 [52] noted that this risk increased even further if the daily consumption of alcohol was greater than two drinks. For example the risk went to a 75% increase when women consumed four or more drinks per day. Alcohol is theorized to cause breast cancer by raising a woman's estrogen levels.
Vitamin A:
Colditz et al [42] noted that the women in the Nurses' Health study who took more Vitamin A were noted to have 22% less breast cancer than women who took smaller or no doses of Vitamin A. (Note: taking more than 10,000 units of Vitamin A during pregnancy may increase the risk of birth defects [53]).
Obesity in postmenopausal women:
Fat (adipose) tissue converts a hormone produced by the adrenal gland called androstenedione to estrone, a type of estrogen. Theoretically, women with higher estrogen levels should have a higher incidence of breast cancer. In a large study of 570,000 Norwegian women Tretli et al [54] found that postmenopausal women who were in the top 25th percentile of weight had an 18% increased risk of breast cancer over women who were at the lower 25th percentile.
History of having had certain “other cancers” in the past:
Women who have or had cancer of the ovary and⁄or uterus appear to be at increased risk of developing breast cancer. A history of ovarian cancer puts a woman at about twice the risk of developing breast cancer, whereas a history of endometrial (uterine) cancer raises the risk by 50% compared to the general population [11]. In addition, having a personal history of malignant melanoma, colon cancer, cancer of the salivary glands, or Hodgkin's lymphoma appears to increase the risk of getting breast cancer in the future [55]. (Conversely, having a history of breast cancer is associated with an increased risk of getting ovarian, uterine, colon or thyroid cancer [55]).
Oncogenes:
Certain people carry genes which may either predispose them to or against breast cancer. BRCA1 and BRCA2 are examples of the former and are thought to make up 80% of the “gene–linked” breast cancer cases. Usually women inherit a normal BRCA1 and BRCA2 gene. These particular genes — tumor suppressor genes–act like “brakes” against breast cancer. If a woman inherits a defective BRCA1 or BRCA2 gene, she will have a markedly increased risk of developing breast cancer. Women with an abnormal BRCA1 gene have an 85% chance of getting breast cancer during their lifetime (ie, 85 out of 100 will get it during an average woman's life span) and a 60% chance of getting ovarian cancer. Jewish women of Ashkenazic descent (ie, most Jewish women in the U.S.) have a higher incidence of carrying a defective BRCA1 gene. The defective BRCA2 gene also confers an 85% lifetime risk of breast cancer. In fact, women who have a defect of this gene are at much higher risk of developing bilateral premenopausal breast cancer. These women also have an increased risk of ovarian cancer–estimated at 15 to 20% in a lifetime. BRCA2 also confers a 50–fold increased risk of developing breast cancer in the male population so that men carrying the gene have an overall lifetime risk of 6%. In spite of all the news about the discovery of the new genes, it is important to keep in mind that only about 5 to 6% of women with breast cancer are thought to have the type that is caused directly by specific gene defects [56,57].
Radiation:
Women who are exposed to radiation, especially before the delivery of their firstborn child, are at increased risk of developing breast cancer. Sadly, this was proven when researchers found that young women who were exposed to radiation doses of 10 reds or more at the time of the atomic blast in Japan, suffered a far higher incidence of breast cancer as they grew older [58]. The increased incidence of breast cancer was smaller in older women who already had children (because they had more fully matured breast cells). The effects of radiation appear to be permanent and are still apparent for as long as 40 years after exposure [11]. Women who have received routine x–rays are probably at little risk of developing cancer from them because they contain small amounts of radiation (e.g., a chest x–ray contains 0.01 reds) whereas a mammogram contains 0.45 reds (based on data from Paul and Juhl's Radiological Imaging, 1993).
Dense mammography:
A woman's mammogram may vary in the “density of its appearance.” A researcher named Dr. Wolfe [59] has noted that women with extremely dense looking mammograms have more than five times the incidence of cancer than women with breasts whose mammograms show a minimally dense appearance.
DES (Diethylstilbestrol):
This artificial estrogen was given to about 2 million women, especially diabetics, to prevent premature labor and miscarriages in the 1940s and was used until the late 1960s [60]. Overall it increases a woman's risk of breast cancer by 35% [61].
Being a twin:
In one study, Hsieh et al [62, p.1321] noted that women who are twins, especially if their twin was a male, experienced a higher in–utero (ie, “when in the uterus”) estrogen level and have noted a trend toward a slightly increased breast cancer risk in these women.
Smoking:
Smoking does not appear to increase the risk of breast cancer. Several well designed studies have failed to find a link [29, 30, 63, 64, 65, 66, 67, 68]. A single study in the Journal of the National Cancer Institute (May, 1998) has noted that women who carry the BRCA1 or BRCA2 gene mutation may have a decreased risk of breast cancer if they have a history of smoking.
Addendum:
In 1981, Pike et al [21] noted that women who had either an induced abortion or a miscarriage prior to their FFTP had an increased risk of breast cancer. The information given in Table IV of their paper enables one to calculate the odds ratio (estimated relative risk) of a miscarriage prior to a FFTP. The number of women with breast cancer who had a miscarriage prior to their FFTP was 13, and the number of “controls” who had a miscarriage prior to their FFTP was 9. The total number of "cases" who did not have a miscarriage before their FFTP was 139 in addition to the 11 women who had induced abortions prior to their FFTP (ie, the latter did not have a miscarriage prior to their FFTP) or 139 + 11 = 150. The number of “controls” who had no miscarriage prior to a FFTP was 261 (253+8).
Using the equation for estimating the relative risk (RR) given in Chapter 4 we obtain:
Estimated [RR = 13⁄150 divided by 9⁄261 = 2.51] or a 151% increased risk of breast cancer for women who have a miscarriage prior to a first birth.
Next page: References:
Article copyrights are held soley by author.